Purple beans are really quite special. The pods are richly colored and easy to spot among the leaves (which is why they’re my favorite kind of “green” bean—so easy to harvest!), and they’re a beautiful contrast to all the emeralds and chartreuses and hunter greens happening in the garden.
I call them magic beans, and the magic happens when you cook them…

Disclosure: If you shop from my article or make a purchase through one of my links, I may receive commissions on some of the products I recommend.
Where do purple beans get their color?
The intense purple color in string beans and snap beans (like Royal Burgundy and Dragon Tongue), yardlong beans (like Chinese Red Noodle), and hyacinth beans (like Ruby Moon) comes from plant pigments called anthocyanins.
Anthocyanins are water-soluble pigments that also give purple cauliflower, purple artichokes, bluish purple peppers, black carrots, and red cabbage their vibrant colors.
Recommended reading: Ruby Kraut (and Why It’s So Good For You)
(But note that anthocyanins are only responsible for certain reds. Some of the red pigments you see, like those in beets and chard, come from betalains, a class of plant pigments that are only found in Caryophyllales. Other red pigments, like those in red peppers and red tomatoes, come from carotenoids.)
Favorite purple bean varieties: Royal Burgundy bush beans, Dragon Tongue bush beans, Trionfo Violetto pole beans, Rattlesnake pole beans, Chinese Red Noodle yardlong beans
Anthocyanins exist in all tissues of a plant and may appear in the roots, stems, leaves, flowers, and fruits (though not all at once).
With purple beans, the pigments primarily show up in the flowers, which gradually give way to purple-tinged pods.

How and why anthocyanins change color in the garden
If you’ve ever grown purple beans yourself, you may have noticed that their color tends to change slightly from season to season, or they don’t appear the same color as other plants you’ve seen. Purple striations may feature more or less, and one year may yield deep purple pods while the next year’s crop appears lighter.
This natural fluctuation in color occurs because anthocyanins are sensitive to the pH level of the “juice” inside the plant cells (the cell sap). The acidity of the cell sap is dependent on both genetic and environmental factors.
Anthocyanins tend to turn red in acidic soil, blue in neutral soil, and yellow in alkaline soil.
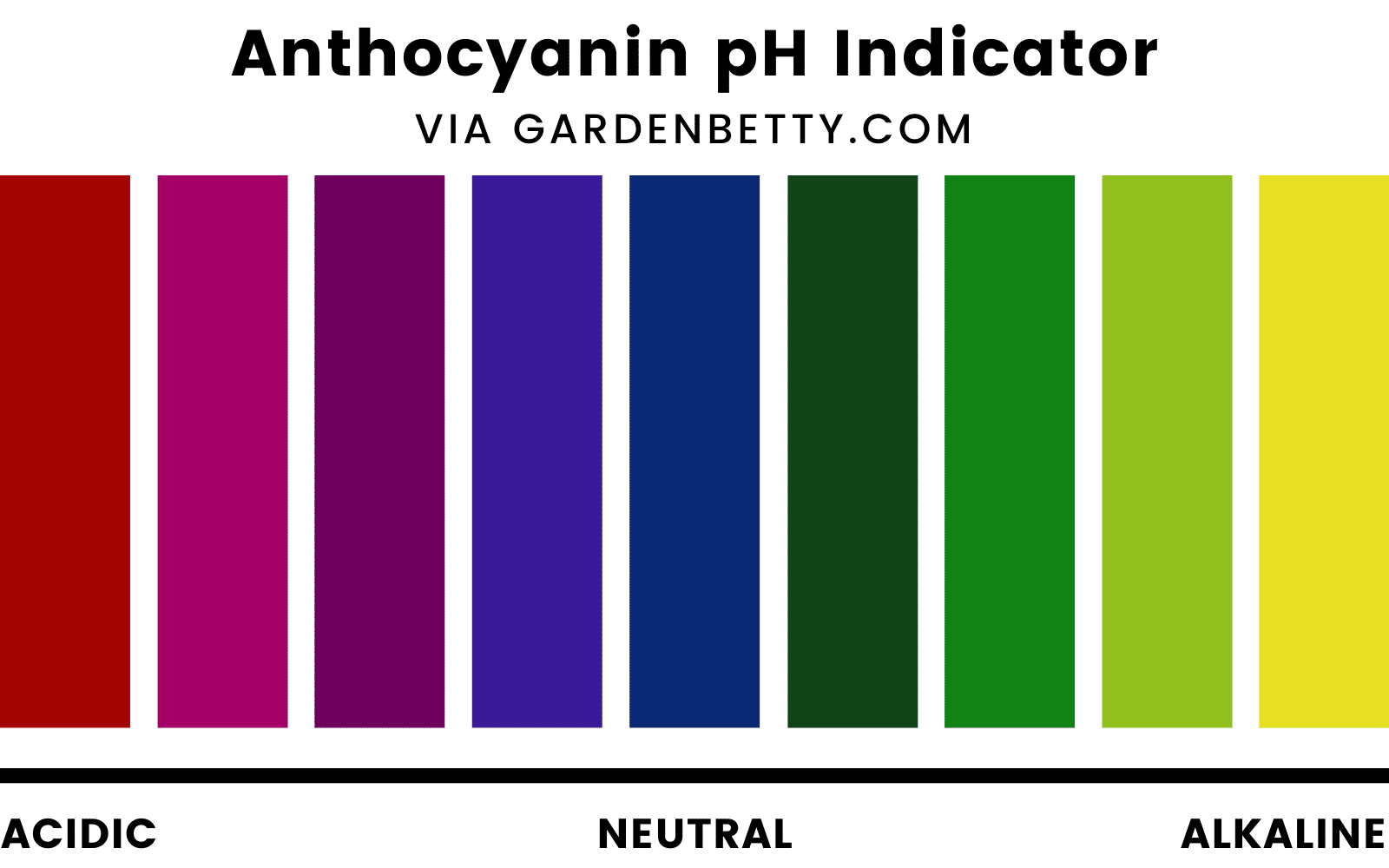
This is why “red” cabbage may appear more purple, and “purple” cauliflower may appear more magenta.
It’s also the reason hydrangeas are famous for their color-shifting ways, as the petals respond highly to varying soil conditions (and gardeners can even change the color of the flowers quite easily by amending their soil to be more or less acidic).
Think of these anthocyanin-rich plants as a litmus test of sorts. (In fact, red cabbage makes a good indicator in pH experiments at home.)

How heat affects anthocyanins
Anthocyanins are highly susceptible to heat and light. You may have seen this anomaly in apples, which sometimes appear more red on one side than the other. This happens when the red side was exposed to more sunshine, spurring a chemical reaction in the plant cells that produces more pigments.
When it comes to purple beans, however, heat plays the principal role. Boiling, baking, or sauteing at high temperatures causes the anthocyanins to deteriorate.
The heat breaks down the plant cells, diluting the acidity of the cell sap as the pigments are dispersed in a more neutral solution (water).
What’s left behind is green chlorophyll, which was always present in the beans but masked by the plant’s anthocyanins.
So, your purple beans end up as green beans and the boiled water looks blue from all the pigments that escaped.

How do you keep purple beans from turning green?
There’s not much you can do to preserve the color if you’re cooking purple beans (and why you don’t see any purple bean casseroles).
If you want to minimize color loss, you can blanch them, singe them on the grill, or toss them into a stir-fry at the end for a couple minutes. The color will fade a bit, but remain purplish.
You can also try to increase the acidity (thus retaining some of the anthocyanins) by briefly soaking the beans in lemon juice or vinegar before cooking them. This trick works best for recipes that already have a bit of tanginess to them, like lemony pastas, Thai-style noodles, or rice bowls with citrus shrimp.
Or, just pick the purple beans when they’re young and tender and enjoy them raw off the vine.
Recipe to try: Three Bean Summer Salad

Why don’t other purple vegetables change color after cooking?
As you may have noticed in the kitchen at one point or another, not all purple vegetables turn green when they’re exposed to heat.
Purple sweet potatoes and purple potatoes retain their vibrant color, in part, because there’s no chlorophyll in the flesh—the tubers grow underground. When they’re cooked (typically by steaming, roasting, or boiling), they may veer more violet since water has a neutral pH, but more or less keep their color.
(Conversely, blue potatoes may take on reddish tones—appearing more purple—if cooked with acidic ingredients.)
But the main reason these tubers—along with certain varieties of purple Brussels sprouts, black carrots, red cabbage, red corn, and red radishes—don’t lose their color is because they contain higher concentrations of acylated anthocyanins.
You see, not all anthocyanins are the same.
In contrast with non-acylated anthocyanins (like those found in purple beans and purple peas), acylated anthocyanins have higher heat and light stability, and lower sensitivity to pH changes.
When these vegetables are cooked past the stage of being crisp-tender, they’ll only fade from purple to lavender because the acylated anthocyanins don’t undergo as drastic of a change.
Acylated anthocyanins are so stable, in fact, that their pigments have been studied as a potential (and more natural and healthful) replacement for synthetic dyes such as FD&C Red No. 40.

The drawback of cooking purple beans
So is there a benefit to preserving the purple color in your beans?
All anthocyanins are tasteless, so they have no effect on flavor. But cooking your purple beans does destroy some of the anthocyanins available.
You’ll reap the most nutritional benefits by eating them raw or cooking them very lightly. Just pick your purple beans when they’re small and tender for the best flavor and texture. (And while you’re at it, pick some of the leaves too—green bean leaves, on their own, are edible and delicious.)
This post updated from an article that originally appeared on July 17, 2013.
View the Web Story on why purple beans turn green after cooking.
















Thanks for solving the magic bean color changing mystery for me! I always wanted to know why this happened
Regards
Judy P
Hey,
thank you for this very interesting article. I grow purple beans and have kinda sad to see that stunning colour simply fade away when cooking them- I now just use them raw in salads.
Interesting about the soil acidity affecting the colour,, I am wondering if I acidify my soil a bit with say sulphur if I will get lovely red beans,, possibly a try for next season!
I found your page yesterday when I was looking for an all natural insecticidal soap for “onion aphids” on my green onions. Today, I learned all about why my purple beans will be purple and how they’ll change when cooking! I grow green beans every year and I always miss some when picking because they blend in so I’m looking forward to an easier harvest this year due to their contrasting color! You give great, easy to understand info, thank you!
Purple potatoes don’t turn green after cooking.
Much as I love purple beans and would like them to retain their beauty … aren’t beans poisonous when raw?
Definitely not, as I have a three-bean salad recipe on this blog that’s delicious! 🙂 http://gardenbetty.com/2012/08/three-bean-summer-salad/
Green beans (aka string beans, snap beans, haricots verts) are not poisonous. They are perfectly safe to eat raw.
On the other hand, red kidney beans should never be consumed raw because they contain extremely high amounts of lectin (a naturally occurring compound in all beans) that are mainly found in the seeds. Just a handful of raw red kidney beans could lead to food poisoning, and while this is alarming, it’s not really an issue since kidney beans are always cooked before serving. Just make sure they’re fully cooked if you’re making them at home.
I just cooked some for the first time and they did not turn green. I overcooked them a bit because I kept waiting for the green! Any idea why they stayed purple?
I’ve never come across a purple bean that didn’t change color after cooking. What variety are yours? And what color are they on the inside when raw?
I bought them at a local farmers market in Atlanta, purple and long, not sure the variety name. The farmer said they would turn green when cooked. They were green on the inside. Weird. I cooked them with some portobello mushrooms.
Maybe a substance in the mushrooms or the cooking liquid, or even the pan you used, inhibited them from losing color. It’s a mystery!
That’s what I’m thinking, that it must have been the mushrooms. I think the cooking liquid was olive oil. Next time I will try steaming them.
Oh, the pan was non-stick Calphalon. I did also add a vegan mushroom gravy.
But why is the cooking water green?
The color in the water comes from pigments that are leached from the beans after cooking. This color actually varies depending on the variety of bean.
Fascinating! My family just planted purple beans for the first time this year and we’re loving the vibrant shade.
The color also makes it much easier to harvest! I don’t know how many green beans I’ve left on my plants until it was too late, just because I didn’t see them.
Are there any health benefits to eating them in their more colorful form, or is it better to get them to a less acidic state?
The health benefit to eating green beans raw (or cooked very minimally) is that you retain more of their nutrients. Plant acidity is not an issue.
TY for RT! @mrswages: RT @theGardenBetty: Blogged on Garden Betty: Why Do Purple Beans Turn Green After #Cooking? http://t.co/lxOvWfpqbr
Blogged on Garden Betty: Why Do Purple Beans Turn Green After Cooking? http://t.co/lxOvWfpqbr < TY for RT! @Angry_Saguaro
@FullCircle05 found this purple bean explanation: be enlightened. http://t.co/GdZKAmEiLD
One of the fascinating things about nature: Why Do Purple Beans Turn Green After Cooking? http://t.co/Dhr47ImI2B #gardenchat #gardening
The short answer: Anthocyanins. The long answer: Why Do Purple Beans Turn Green After Cooking? http://t.co/6joWj4b5eo #gardening #gardenchat
The reason you don’t see any purple bean casseroles! Why Do Purple Beans Turn Green After Cooking? http://t.co/tVYZahtrsJ #gardenchat
Is this the same pigment that’s in purple basil? I’ve always wondered why some of my basils are more purple than others, and why they turn partially green as the season progresses.
Yes, anthocyanins are also the purple pigments found in purple basil (throughout the season, my basil shifts between different shades of purple and green).
The science behind the Royal Burgundy variety: Why Do Purple Beans Turn Green After Cooking? http://t.co/znBTkNY6Ei #gardenchat #gardening
Amy Goo liked this on Facebook.
Amy Watkins Pellicer liked this on Facebook.
Jun Iguchi liked this on Facebook.
Thank you for this post! I’ve been planning on growing these beauties for next summer, so now I think my info is complete!
Royal Burgundy beans are one of my favorites; I grow them every summer!
Just learned something. | Why Do Purple Beans Turn Green After Cooking? http://t.co/jXgjAKHxKb via @zite
RT @theGardenBetty: Blogged on Garden Betty: Why Do Purple Beans Turn Green After #Cooking? http://t.co/cKJiJlGkZZ
RT @theGardenBetty: Blogged on Garden Betty: Why Do Purple Beans Turn Green After Cooking? http://t.co/lxOvWfpqbr
April Leamon liked this on Facebook.
Angela Collins liked this on Facebook.
Marlene Davis Stewart liked this on Facebook.
Why Do Purple Beans Turn Green After Cooking?:
There’s something so majestic about purple beans. The pods are… http://t.co/ROOFWuL9Q5
Blogged on Garden Betty: Why Do Purple Beans Turn Green After Cooking? http://t.co/lxOvWfpqbr